- HOME
- Members
- Principal Investigators
- Aiko Sada
RESEARCH
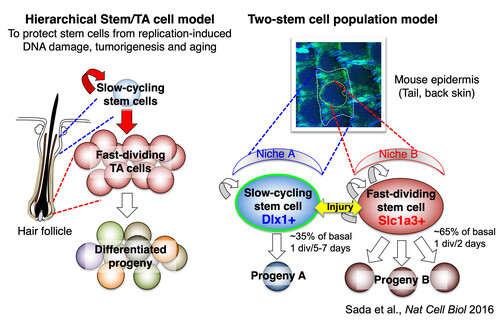
Defining Stem Cell Lineages in the Mouse Skin Epidermis
The skin barrier function is achieved by the proliferation and differentiation of stem cells residing in the epidermis. Epidermal stem cells have been used for clinical purposes since the 1980s, yet their basic knowledge lagged behind until recently due to the lack of molecular markers and tools to define "stem cell" populations. A classical model suggests that slow-cycling cells act as long-lived stem cells and are placed at the top of the lineage hierarchy, generating short-lived progenitor cells. The slow-cycling nature is considered to be a mechanism to protect stem cells from replication-induced DNA damage, accumulation of mutations, and telomere-shortening, all of which can lead to tumorigenesis and aging. However, the true biological functions of slow-cycling cells remain controversial. In our previous study, Sada et al. identified novel molecular markers that enable us to distinguish slow-cycling and fast-dividing cells in the skin epidermis and, thereby, study their localization, kinetics, and long-term stem cell potential during homeostasis and regeneration. Contradicting the classical model, we found that not only slow-cycling cells but also fast-dividing cells act as long-lived stem cells. Each stem cell population clusters within spatially distinct territories in the skin and can preferentially produce its unique lineages. Following injury, these two stem cell populations can interconvert with each other, demonstrating their plasticity for robust skin regeneration. Our work re-defines the stem cell populations in the skin epidermis and identifies two discrete stem cells with different cell division frequencies. This opens a new avenue for understanding the heterogeneous stem cell populations in aging, cancer, and other diseases.
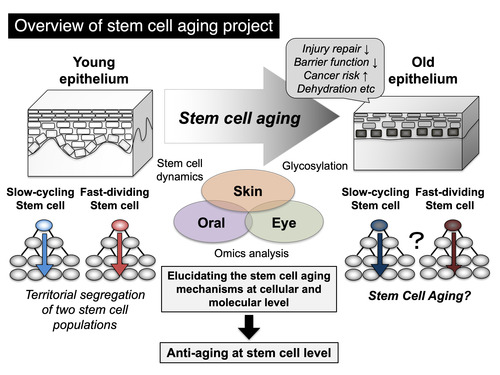
Elucidating the Mechanisms of Stem Cell Aging
Aging is a gradual decline in physiological functions over a course of time. It still remains largely unknown what are the crucial drivers for aging at cellular and molecular levels. A theory of "stem cell aging" suggests that aging is caused by misregulation or dysfunction of stem cells in aged tissues. The aged epithelial tissues show reduced wound healing and barrier function, with an increased risk of cancer. Our recent study proposed the co-existence of two distinct stem cell populations - slow-cycling and fast-dividing stem cells - in the mouse epidermis; however, it remains largely unknown how aging affects these stem cell populations and how it contributes to age-associated tissue dysfunction. Considering that fast-dividing stem cells experience three times more divisions than slow-cycling stem cells over the entire life span, they may lose their stem cell ability prematurely or cause malignant transformation due to life-long, repeated cell divisions. Our research aims to understand the cellular and molecular basis of stem cell aging in three epithelial tissues, skin, oral, and eyes, with implications for future treatments of age-related disorders including cancer.