- HOME
- News & Events
- Publications
- Activation of Transforming Growth Factor Beta 1 Signaling in Gastric Cancer-associated Fibroblasts I...
Publications
Activation of Transforming Growth Factor Beta 1 Signaling in Gastric Cancer-associated Fibroblasts Increases Their Motility, via Expression of Rhomboid 5 Homolog 2, and Ability to Induce Invasiveness of Gastric Cancer Cells
July 5 2017
Takatsugu Ishimoto
Paper information
Takatsugu Ishimoto#, Keisuke Miyake#, Tannistha Nandi#, Masakazu Yashiro, Nobuyuki Onishi, Kie Kyon Huang, Suling Joyce Lin, Ramnarayanan Kalpana, Su Ting Tay, Yuka Suzuki, Byoung Chul Cho, Daisuke Kuroda, Kota Arima, Daisuke Izumi, Masaaki Iwatsuki, Yoshifumi Baba, Eiji Oki, Masayuki Watanabe, Hideyuki Saya, Kosei Hirakawa, Hideo Baba* and Patrick Tan*
(#These authors contributed equally to this work, *corresponding authors)
Activation of Transforming Growth Factor Beta 1 Signaling in Gastric Cancer-associated Fibroblasts Increases Their Motility, via Expression of Rhomboid 5 Homolog 2, and Ability to Induce Invasiveness of Gastric Cancer Cells.
Gastroenterology. 2017 Jul;153(1):191-204.e16. doi: 10.1053/j.gastro.2017.03.046.
Highlights
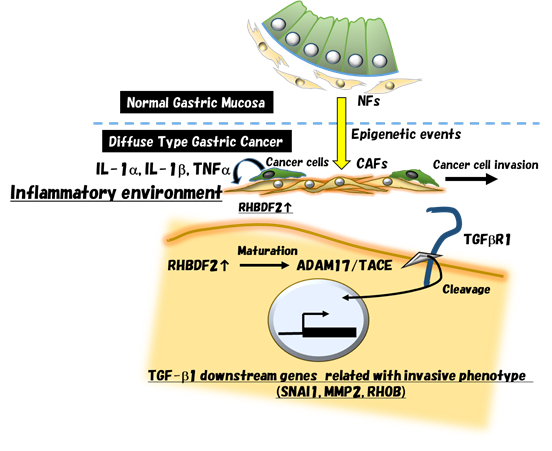
l CAFs exhibit an invasive molecular pattern and high motility in the ECM
l High-motility CAFs confer on DGC cells the ability to invade the ECM
l RHBDF2 is up-regulated by pro-inflammatory cytokines and activates TACE
l Expression of RHBDF2-inducing cytokines predicts survival probability in GC patients
Abstract
Cancer-associated fibroblasts (CAFs) are intimately associated with cancer cells and promote tumor progression in a wide variety of epithelial malignancies. Here, we show that RHBDF2 promotes both the cleavage of type I transforming growth factor (TGF)-b receptor through tumor necrosis factor α converting enzyme activation and the motility of CAFs upon stimulation with TGF- b 1. RHBDF2 expression is up-regulated by pro-inflammatory cytokines during chronic interactions between stromal fibroblasts and diffuse-type gastric cancer cells. Consequently, high-motility CAFs confer on cancer cells the ability to invade the extracellular matrix. Together, the gene expression alterations in stromal fibroblasts caused by neighboring cancer cells give rise to CAFs exhibiting an invasive molecular pattern, which, in turn, support cancer cell invasion.