- HOME
- News & Events
- Publications
- 【Publications】Exposure to microbial products followed by loss of Tet2 promotes myelodysplastic syndr...
Publications
【Publications】Exposure to microbial products followed by loss of Tet2 promotes myelodysplastic syndrome via remodeling HSCs
May 30 2023
Lab: Goro Sashida
Paper information
Tile:
Exposure to microbial products followed by loss of Tet2 promotes myelodysplastic syndrome via remodeling HSCs
Takako Yokomizo-Nakano, Ai Hamashima, Sho Kubota, Jie Bai, Supannika Sorin, Yuqi Sun, Kenta Kikuchi, Mihoko Iimori, Mariko Morii, Akinori Kanai, Atsushi Iwama, Gang Huang, Daisuke Kurotaki, Hitoshi Takizawa, Hirotaka Matsui, Goro Sashida
Journal of Experimental Medicine 2023 Apr 220(7) e20220962 doi: 10.1084/jem.20220962
Highlights
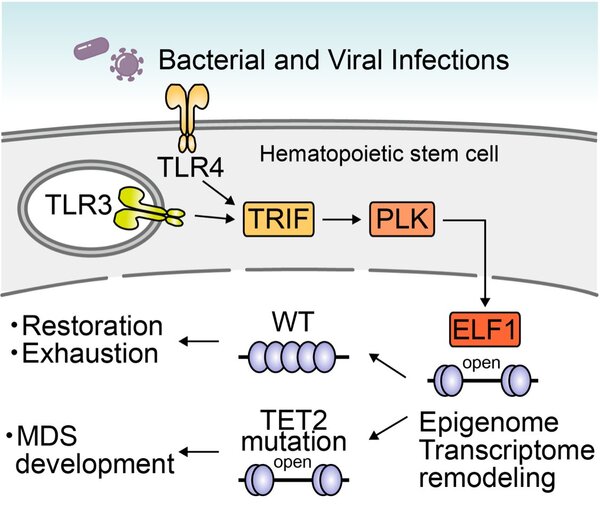
- Prior infection stress followed by the loss of Tet2 remodeled the epigenome and transcriptome in HSCs via the Tlr3/4-Myd88/Trif-Plk-Elf1 axis, which drove the development of MDS.
- The present study proposes the Trif-Plk-Elf1 axis as a potential target for therapeutic interventions for MDS.
Abstract:
Aberrant innate immune signaling in myelodysplastic syndrome (MDS) hematopoietic stem/progenitor cells (HSPCs) has been implicated as a driver of the development of MDS. We herein demonstrated that a prior stimulation with bacterial and viral products followed by loss of the Tet2 gene facilitated the development of MDS via up-regulating the target genes of the Elf1 transcription factor and remodeling the epigenome in hematopoietic stem cells (HSCs) in a manner that was dependent on Polo-like kinases (Plk) downstream of Tlr3/4-Trif signaling, but did not increase genomic mutations. The pharmacological inhibition of Plk function or the knockdown of Elf1 expression was sufficient to prevent the epigenetic remodeling in HSCs and diminish the enhanced clonogenicity and the impaired erythropoiesis. Moreover, this Elf1-target signature was significantly enriched in MDS HSPCs in humans. Therefore, prior infection stress and the acquisition of a driver mutation remodeled the transcriptional and epigenetic landscapes and cellular functions in HSCs via the Trif-Plk-Elf1 axis, which promoted the development of MDS.
Graphical abstract