- HOME
- News & Events
- Publications
- 【Publications】IL-1β derived from mixed-polarized macrophages activates fibroblasts and synergistical...
Publications
【Publications】IL-1β derived from mixed-polarized macrophages activates fibroblasts and synergistically forms a cancer-promoting microenvironment
December 15 2022
Lab:Takatsugu Ishimoto
Paper information
Tile:
IL-1β derived from mixed-polarized macrophages activates fibroblasts and synergistically forms a cancer-promoting microenvironment
Jun Zhang, Lingfeng Fu, Noriko Yasuda-Yoshihara, Atsuko Yonemura, Feng Wei, Luke Bu, Xichen Hu, Takahiko Akiyama, Fumimasa Kitamura, Tadahito Yasuda, Takashi Semba, Tomoyuki Uchihara, Rumi Itoyama, Kohei Yamashita, Kojiro Eto, Shiro Iwagami, Masakazu Yashiro, Yoshihiro Komohara, Hideo Baba*, Takatsugu Ishimoto*
(*corresponding authors)
Gastric Cancer 2022 Dec. doi: 10.1007/s10120-022-01352-3
Highlights
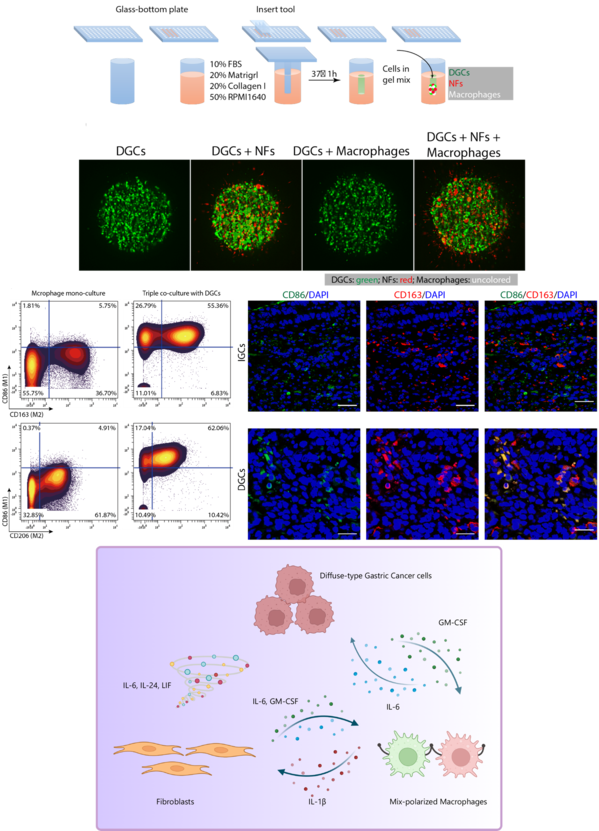
- The original 3D triple coculture system determined the promotional effects of nonmalignant cells on DGC invasive growth.
- We identified mix-polarized macrophages with M1/M2 cell surface markers in triple coculture and human DGC tissues.
- IL-1β from mix-polarized macrophages induced the conversion of NFs into cancer-associated fibroblast-like (CAF-like) cells.
- IL-6 and colony stimulating factor 2 (GM-CSF) cooperated to maintain the stable state of mix-polarized macrophages.
Abstract
Remodeling of tumor microenvironment (TME) to benefit cancer cells is crucial for tumor progression. Although diffuse-type gastric cancer (DGC) preferentially interacts with the TME, the precise mechanism of complicated network remains unknown. This study aimed to investigate the mutual activation mechanism underlying DGC progression. Cohort analysis revealed that DGC patients had a poor prognosis. The noncancerous fibroblasts (NFs) and macrophages interacted with DGC cells to form a cell cluster in the invasive front of DGC tissue. Original 3D triple co-culture system determined the promotional effects of nonmalignant cells on DGC invasive growth. Mass cytometry analysis of co-cultured macrophages, NFs, and DGC cells was performed. We notably identified a mix-polarized macrophage cell type with M1/M2 cell surface markers in triple co-culture system. IL-1β from mix-polarized macrophages induced the conversion of NFs to cancer-associated fibroblast-like (CAF-like) cells, promoting the malignant phenotype of DGC cells by inducing the secretion of IL-6, IL-24, and leukemia inhibitory factor (LIF). Moreover, IL-6 and colony stimulating factor 2 (GM-CSF) cooperated to maintain the stable state of mix-polarized macrophages. Finally, the TCGA and Kumamoto University cohorts were used to evaluate the clinical relevance of the in vitro findings. We found that mix-polarized macrophages were frequently detected in DGC tissues. These findings demonstrated that mix-polarized macrophages exist as a novel subtype through the reciprocal interaction between DGC cells and nonmalignant cells.
Representative Figure