- HOME
- News & Events
- Publications
- 【Publications】CD271+CD51+PALLADIN- human mesenchymal stromal cells possess enhanced ossicle-forming ...
Publications
【Publications】CD271+CD51+PALLADIN- human mesenchymal stromal cells possess enhanced ossicle-forming potential
May 7 2021
Lab: Hitoshi Takizawa
Paper information
Title:
CD271+CD51+PALLADIN- human mesenchymal stromal cells possess enhanced ossicle-forming potential
Maiko Sezaki, Biswas Subinoy, Sayuri Nakata, Motohiko Oshima, Shuhei Koide, Nicole Pui Yu Ho, Nobukazu Okamoto, Takeshi Miyamoto, Atsushi Iwama and Hitoshi Takizawa
Stem Cells and Development. ahead of print
Online Ahead of Editing: April 30, 2021
doi: http://doi.org/10.1089/scd.2021.0021
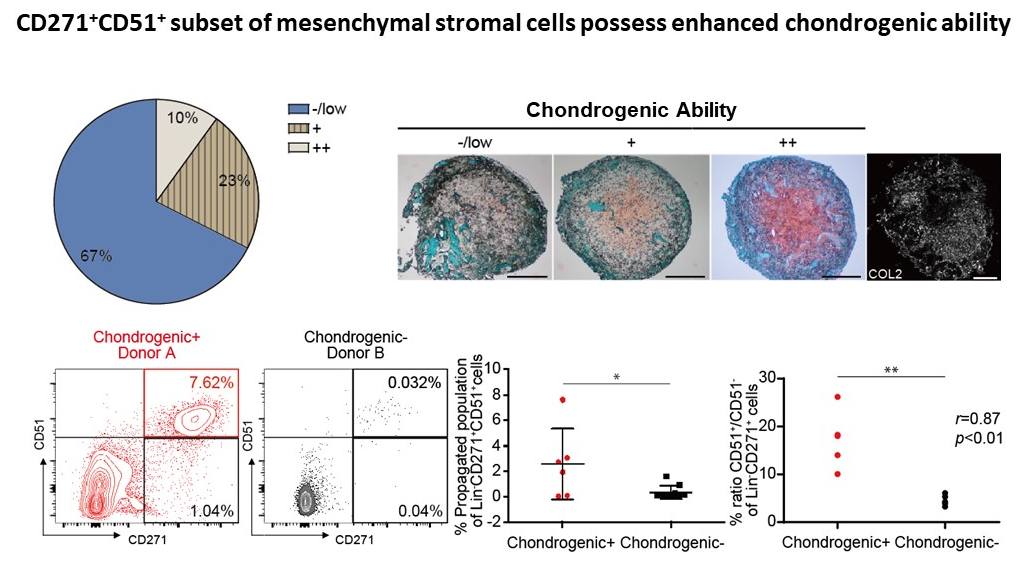
- Only 30% of human BM donors are able to form ectopic bone organs
- Chondrogenic ability is enriched in the CD271+CD51+ subset of MSCs
- PALLADIN expression better defines MSCs with chondro/osteogenesis potential
Abstract
Human mesenchymal stem/stromal cells (hMSCs), when engrafted into immunodeficient mice can form ectopic bone organs with hematopoietic stem cell supportive functions. However, the ability to do so, through a cartilage intermediate appears limited to 30% of donor bone marrow samples. Here, we characterize the heterogeneous nature of hMSCs and their ability to efficiently form humanized ossicles observed in "good donors" to correlate with the frequency and functionality of chondrocyte progenitors. Flow cytometry of putative hMSC markers were enriched in the CD271+CD51+ stromal cell subset, which also possessed enhanced hMSC activity as assessed by single-cell CFU-F and undifferentiated mesensphere formation. Transcriptome analysis of CD271+ cells presented upregulation of chondro/osteogenesis-related genes and those related to HSC/niche maintenance factors such as CXCL12 and ANGIOPOIETIN 1. Among the candidate genes selected to enrich for subsets with greater chondrogenic ability, cells negative for the actin cross-linker, PALLADIN displayed the greatest CFU-F potential. Our study contributes to a better characterization of ossicle-forming hMSCs and their efficient isolation for the optimized engineering of human bone organs.
(Click for a larger image)