- HOME
- News & Events
- Publications
- 【Publications】Autophagy is dispensable for the maintenance of hematopoietic stem cells in neonates
Publications
【Publications】Autophagy is dispensable for the maintenance of hematopoietic stem cells in neonates
March 19 2021
Lab: Toshio Suda
Paper information
Michihiro Hashimoto*, Terumasa Umemoto*, Ayako Nakamura-Ishizu, Takayoshi Matsumura, Tomomasa Yokomizo, Maiko Sezaki, Hitoshi Takizawa, Toshio Suda
*These authors have contributed equally to this work.
Title
Autophagy is dispensable for the maintenance of hematopoietic stem cells in neonates
Blood Adv 2021; 5 (6): 1594-1604.
doi: https://doi.org/10.1182/bloodadvances.2020002410
Key Points
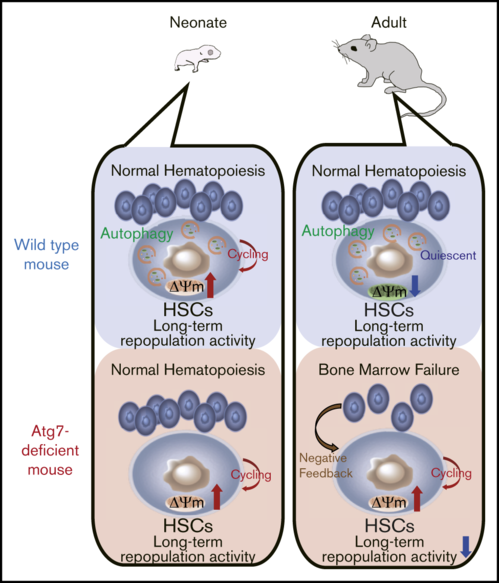
- Atg7 deficiency impairs HSC quiescence through dysregulation of mitochondrial metabolism, resulting in bone marrow failure at adult stage.
- Atg7-deficient neonatal HSCs normally show long-term engraftment, which suggests Atg7-independent hematopoiesis.
Abstract
Hematopoietic stem cells (HSCs) undergo self-renewal or differentiation to sustain lifelong hematopoiesis. HSCs are preserved in quiescence with low mitochondrial activity. Recent studies indicate that autophagy contributes to HSC quiescence through suppressing mitochondrial metabolism. However, it remains unclear whether autophagy is involved in the regulation of neonatal HSCs, which proliferate actively. In this study, we clarified the role of autophagy in neonatal HSCs using 2 types of autophagy-related gene 7 (Atg7)-conditional knockout mice: Mx1-Cre inducible system and Vav-Cre system. Atg7-deficient HSCs exhibited excess cell divisions with enhanced mitochondrial metabolism, leading to bone marrow failure at adult stage. However, Atg7 deficiency minimally affected hematopoiesis and metabolic state in HSCs at neonatal stage. In addition, Atg7-deficient neonatal HSCs exhibited long-term reconstructing activity, equivalent to wild-type neonatal HSCs. Taken together, autophagy is dispensable for stem cell function and hematopoietic homeostasis in neonates and provide a novel aspect into the role of autophagy in the HSC regulation.