- HOME
- News & Events
- Publications
- 【Publications】Colorectal cancer stem cells acquire chemoresistance through the upregulation of FBXW7...
Publications
【Publications】Colorectal cancer stem cells acquire chemoresistance through the upregulation of FBXW7 and the consequent degradation of c-Myc
August 1 2017
Takatsugu Ishimoto
Paper information
Izumi D, Ishimoto T*, Miyake K, Eto T, Arima K, Kiyozumi Y, Uchihara T, Kurashige J, Iwatsuki M, Baba Y, Sakamoto Y, Miyamoto Y, Yoshida N, Watanabe M, Goel A, Tan P, Baba H
(*corresponding author)
Colorectal cancer stem cells acquire chemoresistance through the upregulation of FBXW7 and the consequent degradation of c-Myc.
Stem Cells. 2017 Jul 11. doi: 10.1002/stem.2668. [Epub ahead of print].
Highlights
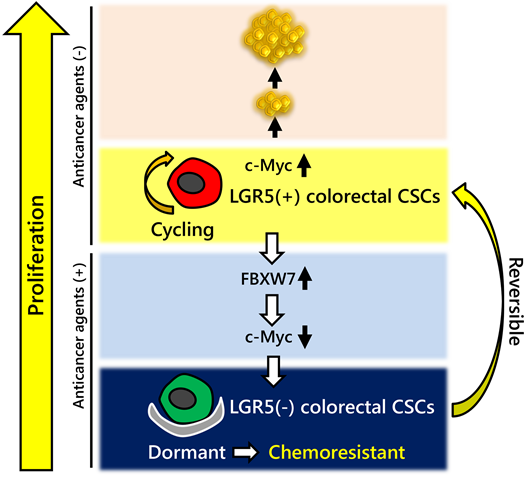
-c-Myc expression is downregulated in LGR5-drug-resistant colorectal CSCs
-FBXW7 is a c-Myc protein regulator in colorectal CSCs during exposure to anticancer agents
- Colorectal CSCs acquire chemoresistance by upregulating FBXW7 expression
- FBXW7 regulates a CSC-specific drug resistance mechanism
- FBXW7 is highly expressed in liver metastatic colorectal cancer tissues following chemotherapy
Abstract
The cancer stem cell (CSC) paradigm suggests that tumors are organized hierarchically. We previously established an LGR5+ human colorectal cancer stem-cell-enriched cell line (colorectal CSCs) that expresses well-accepted colorectal CSC markers and that can dynamically switch between proliferative and drug-resistant non-cycling states. We performed this study to elucidate the molecular mechanisms responsible for evading cell death in colorectal CSCs mediated by anticancer agents. During the cell cycle arrest caused by anticancer agents, we found that c-Myc expression was substantially decreased in colorectal CSCs. The c-Myc expression alterations were mediated by upregulation of FBXW7, as evidenced through FBXW7-siRNA knockdown experiments that resulted in enhanced cell sensitivity to anticancer agents. Upregulation of FBXW7 following drug treatment was not evident in commercially available cancer cell lines. Colorectal CSCs were induced to differentiation by Matrigel and fetal bovine serum. Differentiated CSCs treated with anticancer agents did not show upregulation of FBXW7 and were more sensitive to CPT-11, highlighting the potential CSC-specific nature of our data. The FBXW7 over-expression was further validated in resected liver metastatic sites in colorectal cancer patients after chemotherapy. In conclusion, our study revealed that a CSC-specific FBXW7-regulatory mechanism is strongly associated with resistance to chemotherapeutic agents. Inhibition of FBXW7-upregulation in CSCs following chemotherapy may enhance the response to anticancer agents and represents an attractive strategy for the elimination of colorectal CSCs.