- HOME
- News & Events
- Publications
- 【Publications】Integrin αvβ3 enhances the suppressive effect of interferon-γ on hematopoietic stem ce...
Publications
【Publications】Integrin αvβ3 enhances the suppressive effect of interferon-γ on hematopoietic stem cells
July 3 2017
Terumasa Umemoto
Paper information
Terumasa Umemoto*, Yu Matsuzaki, Yoshiko Shiratsuchi, Michihiro Hashimoto, Takayuki Yoshimoto, Ayako Nakamura-Ishizu, Brian Petrich, Masayuki Yamato and Toshio Suda*
(*corresponding authors)
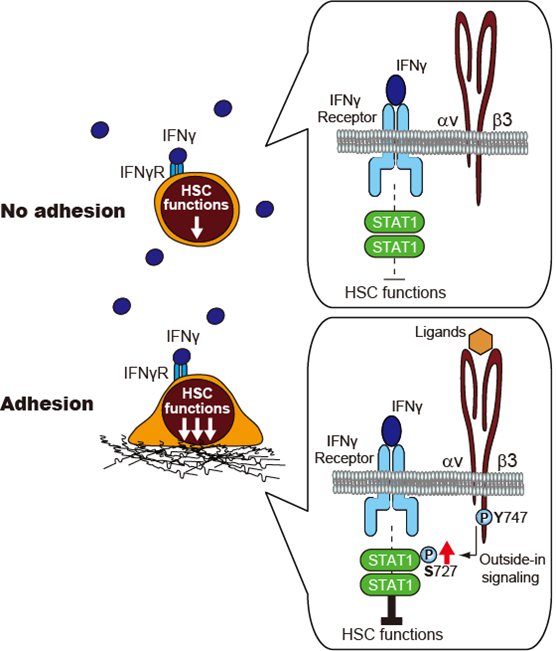
Although integrin β3 signaling contributes to the maintenance of hematopoietic stem cells (HSCs), this integrin signaling exerts the synergistic negative regulation of HSCs in the context that pro-inflammatory cytokine interferon-γ (IFNγ) suppresses HSC functions. Thus, β3 integrin signaling-mediated regulation of HSCs is context-dependent.
EMBO J, 2017, Jul 3. pii: e201796771. doi: 0.15252/embj.201796771
Highlights
-Impaired integrin β3 signaling mitigates IFNγ-dependent negative action on HSCs.
-During IFNγ stimulation, the induction of integrin β3 signaling intensifies the suppressive effect of IFNγ on HSCs.
-In the absence of IFNγ, the induction of integrin β3 signaling upregulates HSC activity.
-During IFNγ stimulation, integrin β3 signaling enhances STAT1-mediated gene expression via serine phosphorylation
Abstract
Hematopoietic homeostasis depends on the maintenance of hematopoietic stem cells (HSCs), which are regulated within a specialized bone marrow (BM) niche. When HSC sense external stimuli, their adhesion status may be critical for determining HSC cell fate. The cell surface molecule, integrin αvβ3 is activated through HSC adhesion to extracellular matrix and niche cells. Integrin β3 signaling maintains HSCs within the niche. Here, we showed the synergistic negative regulation of the pro-inflammatory cytokine interferon-γ (IFNγ) and β3 integrin signaling in HSC function by a novel definitive phenotyping of HSCs. Integrin αvβ3 suppressed HSC function in the presence of IFNγ, and impaired integrin β3 signaling mitigated IFNγ-dependent negative action on HSCs. During IFNγ stimulation, integrin β3 signaling enhanced STAT1-mediated gene expression via serine phosphorylation. These findings show that integrin β3 signaling intensifies the suppressive effect of IFNγ on HSCs, which indicates that cell adhesion via integrin αvβ3 within the BM niche acts as a context-dependent signal modulator to regulate the HSC function under both steady state and inflammatory conditions.