Motomi Osato
Paper Information
Wang QC, Chin DWL, Chooi JY, Chng WJ, Taniuchi I, Tergaonkar V, Osato M.
Cbfb deficiency results in differentiation blocks and stem/progenitor cell expansion in murine hematopoiesis. Leukemia (IF 9.379), 29: 753-57, 2015 PMID: 25371180
http://www.ncbi.nlm.nih.gov/pubmed/25371180
Highlights
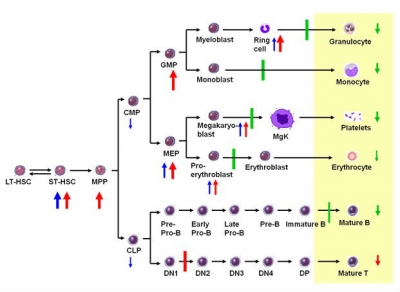
- Cbfb conditional knockout mice exhibit differentiation blocks and stem cell expansion in hematopoiesis.
- Such phenotypes are more pronounced when Cbfb is inactivated at neonatal stage, as compared to those at adult stage.
Abstract
The PEBP2/CBF heterodimeric transcription factors consist of two subunits: a DNA-binding α subunit (Runx1, Runx2 or Runx3) and a non-DNA-binding β subunit (PEBP2b/Cbfβ) encoded by Cbfb. RUNX1 and CBFB play critical roles in hematopoiesis and are frequently mutated in human leukemia. Similar toRunx1-/- mice, Cbfb-/- embryos die at midgestation due to hemorrhage and complete lack of hematopoietic stem cells (HSCs). Due to this embryonic lethality, Cbfb function in adult hematopoiesis remains unknown. Here, we show that Cbfb conditional knockout (cKO) mice exhibited differentiation blockages and HSC expansion that are more drastic than Runx1 cKO mice. Notably, such precancerous status was more pronounced when Cbfb was inactivated at neonatal stage by using Vav-iCre system, as compared to that at adult stage by Mx-Cre system. This age-dependent phenotypic enhancement may underlie the high prevalence of human RUNX leukemia in childhood and younger adults.